On December 24, the AKK AM06™ strain, a new generation of probiotics with independent intellectual property rights of Zhiyi Biopharma/Puwei Junjian, represented by Tech-inno Health, was recognized by the American Expert Committee as Self-affirmed GRAS, marking that the AKK AM06™ strain was officially included in the globally authoritative recognized safety list.
As early as August this year, the two sides have jointly established the "Guangdong Provincial Living Biopharmaceutical Engineering Technology Research Center - Second-Generation Probiotics Innovative Food Collaborative R&D Center" to actively cooperate on projects such as "innovative research and development of second-generation probiotic new food raw materials and products" and "functional food screening and evaluation research". In the future, the two parties will continue to have in-depth exchanges and discuss the research, development and application of AKK AM06™ to provide consumers with more and better second-generation probiotic products.
As the sole distributor of AKK AM06™ strain in the world, Tech-inno Health also looks forward to working with domestic and foreign partners to jointly promote the comprehensive development of this probiotic and bring safer and effective probiotic health solutions to consumers around the world.

About GRAS certification
Authorized by the U.S. Congress, the federal government, the FDA is the highest law enforcement agency specializing in food and drug administration. GRAS, short for "Generally Recognized As Safe", is the FDA's Generally Recognized As Safe (GRAS) scientific evaluation, which requires a rigorous scientific evaluation and review process. GRAS certification standards are extremely strict, covering many key aspects of the product, such as raw material source, production process, and quality control.
The successful GRAS certification of the AKK AM06™ strain is a testament to the product's international standards in terms of safety, compliance and quality. This is a strong signal of trust, and it means that AKK AM06™ has been vetted by one of the world's strictest food safety regulators, ensuring that it is safe for consumers during use.
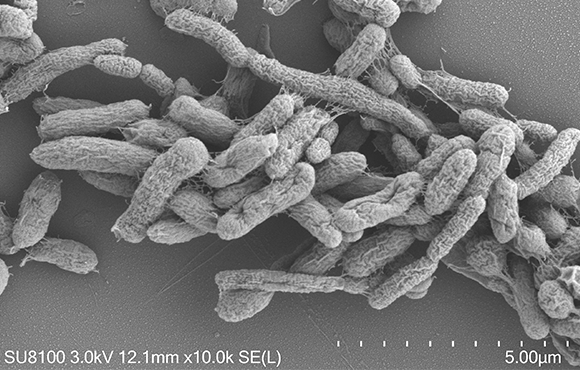
AKK is known as a model of next-generation probiotics and is one of the most promising next-generation probiotics in the international market
AKK is the abbreviation of Akkermansia muciniphila, which was isolated from healthy human feces in 2004 by the team of Professor Willem M. de Vos. AKK bacteria account for 1~5% of the human microbial community, which is a resident bacteria in the human intestine, colonizing the intestinal mucus layer and feeding on intestinal mucin. The abundance of AKK bacteria in the gut is related to the age, diet and health status of the host, and the abundance is generally lower in the Chinese population. A large number of studies have shown that AKK bacteria have obvious effects in regulating the intestinal barrier, weight management, hypoglycemia, anti-aging, etc., and the inactivation of AKK bacteria has the same biological effect as live bacteria, or even better than live bacteria.
AKK AM06™ is currently the only AKK bacteria derived from breast milk, and it has a high safety profile

Studies have shown that probiotics in breast milk are able to be transferred to the infant gut via the oral-gut-mammary pathway, which has a significant effect on infant health, and these strains are natural, evolutionarily adapted, and therefore generally considered safe. In contrast, strains of fecal origin may contain a variety of bacteria (including beneficial bacteria and potentially pathogenic bacteria) that require more rigorous screening and validation due to their more complex composition.
In 2023, the technology development team published a breakthrough study in the journal Microbiology Spectrum. The study performed a comprehensive analysis of 31 isolates of AKK bacteria from healthy individuals (30 from feces and 1 from breast milk). The study found that the AM06™AKK strain derived from healthy breast milk had significant advantages over the AKK strain derived from healthy human feces in terms of low toxicity, high genetic stability, high pH and bile salt tolerance, making it an ideal candidate for probiotic applications in food.
Inactivation of AKK AM06™ strain raw materials, better effect, wider commercial application
AKK AM06™ strain adopts its own PROFORCARE®Heat-InActived inactivation technology, which can completely inactivate AKK bacteria under low temperature conditions, while keeping the bacteria intact without fragmentation, retaining high genetic stability, and more adaptable to the complex environment of the gastrointestinal tract, while effectively retaining the active ingredients of the bacteria. Unlike live bacteria, inactivated probiotics are not affected by stomach acid and can be used with antibiotics, whereas live bacteria should not be taken with antibiotics. These characteristics make inactivated AKK bacteria have a wider commercial application prospect, but also provide consumers with more product choices, and have significant advantages in the field of probiotic applications.
Along the way, Tech-inno Health has always been driven by innovation and fully invested in the deep cultivation of the big health industry. We are not only actively engaged in R&D, but also look at the global market, seeking in-depth cooperation with various partners, aiming to jointly expand the application boundaries of AKK AM06™ in the field of health, provide consumers with more comprehensive and scientific health solutions, and help realize the beautiful vision of national health.
In the future, Tech-inno Health will continue to give full play to its own technological advantages and work with partners to create the future of the health industry, so that more people can enjoy the health benefits brought by probiotics.
About Zhiyi Biotech / Puwei Junjian
Zhiyi Biotech is one of the earliest units to carry out AKK research in China, and has published a number of scientific papers around the AKK AM06™ strain, and has applied for 22 domestic and foreign patents. National Natural Science Foundation projects have carried out basic research on AKK AM06™. As a wholly-owned subsidiary of Zhiyi Biotech, PlusGuardian focuses on the research and development of functional probiotics, has China's leading innovative probiotic R&D and production system, and has built two production bases of 5000L fermenters, with world-class production process capabilities, drug-grade quality control of products, and guaranteed quality, stability and production capacity.